The Canadian natural health product (NHP) industry is undergoing a significant transformation with the introduction of new Plain Language Labelling (PLL) Regulations. These updated guidelines, published by Health Canada in 2022, aim to enhance transparency, standardization, and accessibility in NHP labelling. By replacing the previous 2006 regulations, the PLL framework ensures that consumers can easily understand critical product information, including dosage instructions, warnings, and ingredients.
With the increasing reliance on NHPs for wellness and self-care, clear labelling is essential for consumer safety and informed decision-making. Misinterpretation of product labels has been a long-standing concern, leading to accidental misuse or allergic reactions. The updated regulations directly address these risks by mandating standardized formats, enhanced readability features, and bilingual labelling requirements.
Our guide provides an overview of the key regulatory changes, their impact on consumers and industry stakeholders, and the steps businesses must take to ensure compliance with Health Canada’s revised labelling standards
- Purpose of Plain Language Labelling (PLL)
- Key Updates to Natural Health Product (NHP) Labelling Regulations
- Inner and Outer NHP Labelling Requirements
- Comparison of Previous and Updated Labelling Requirements
- Significance of These Changes for Consumers and Industry Stakeholders
- Compliance Deadlines for Industry Participants
- Guidelines for Achieving Compliance with the Updated Regulations
- Regulatory Oversight and Enforcement by Health Canada
- Final Remarks
- FAQ
Purpose of Plain Language Labelling (PLL)
The purpose of the PLL is to enhance the accessibility and clarity of health product information for all Canadians. These label requirements are designed to ensure that consumers can easily interpret details such as dosage, potential risks, and ingredients, thereby reducing the risk of misuse or adverse reactions. Under the new requirements, NHP labels must present information in a standardized format using clear, concise language and enhanced readability features.
Benefits of the Product Facts Table (PFT)
Standardizing NHP labelling allows consumers to efficiently compare product information across different brands, catering to their needs while ensuring key details are easily accessible. Previously, the 2006 labelling regulations did not require a standardized table format, permitting manufacturers to present information in various ways. While this flexibility benefited marketing and brand identity, it lacked uniformity, requiring consumers to spend additional time reviewing labels to understand a product’s intended use and cautionary statements. The updated regulations address these challenges by introducing a clear and consistent approach to product labelling.
Key Updates to Natural Health Product (NHP) Labelling Regulations
The new labelling requirements incorporate several substantial changes to enhance clarity and uniformity in labelling practices for NHP labels, including:
Standardized Product Facts Table (PFT)
The updated regulations for NHPs now require the inclusion of a standardized Product Facts Table (PFT) on product labels to enhance clarity, consistency, and consumer understanding. This table must present essential details such as medicinal ingredients, intended uses, warnings, dosage instructions, storage guidelines, non-medicinal ingredients, and manufacturer contact information.
To ensure uniformity and readability, Health Canada has outlined specific design and formatting requirements, including the use of a sans-serif font with a minimum size, high-contrast colours, and clearly defined borders and line separations. The PFT must also be bilingual, with the option of a single bilingual table or two separate tables for English and French.

While most NHPs must comply with these new standards, certain exemptions apply to products with small packaging, short-term use items, and those with localized effects, such as topical treatments. In cases where label space is insufficient, manufacturers may use fold-out labels, inserts, or direct consumers to online resources.
Mandatory Allergen Labelling
In addition to PFT requirements, manufacturers must clearly declare common food allergens such as nuts, dairy, wheat, and shellfish within the Warnings section to ensure they are easily visible to consumers. This includes not only direct allergens, but also ingredients derived from allergenic sources, gluten sources, and added sulphites where present. For example, if a product contains whey protein, “milk” must be explicitly identified as the allergen. These requirements align with broader Canadian labelling frameworks and are intended to support transparency and reduce the risk of unintended allergen exposure.
Enhanced Readability Standards – Font and Colour Requirements
The labelling update also introduces strict formatting and typographic standards to ensure that labels are clear, readable, and easy for consumers to navigate.
New Font Size Requirements
The regulations also establish specific font size requirements depending on the type of information being displayed. For instance, medicinal ingredients must appear in at least a 6-point font, or 5.5 points if using a condensed typeface. Non-medicinal ingredients must be presented in a minimum 5.5-point font, or 5 points for condensed fonts. These minimum sizes prevent manufacturers from using overly small text that could compromise readability.
Requirements for Bolded Titles and Statements
To further improve clarity, the rules specify that headings and subheadings within the PFT must be bolded, distinguishing them from the body text, which remains in a regular-weight font. This structured formatting ensures a logical flow of information, making it easier for consumers to locate key details.
The PFT must contain the following elements in bold:
- Table title “Product Facts”
- Headings “Use,” “Warnings,” etc.
- Subheadings “Do not use,” “Keep out of reach of children”
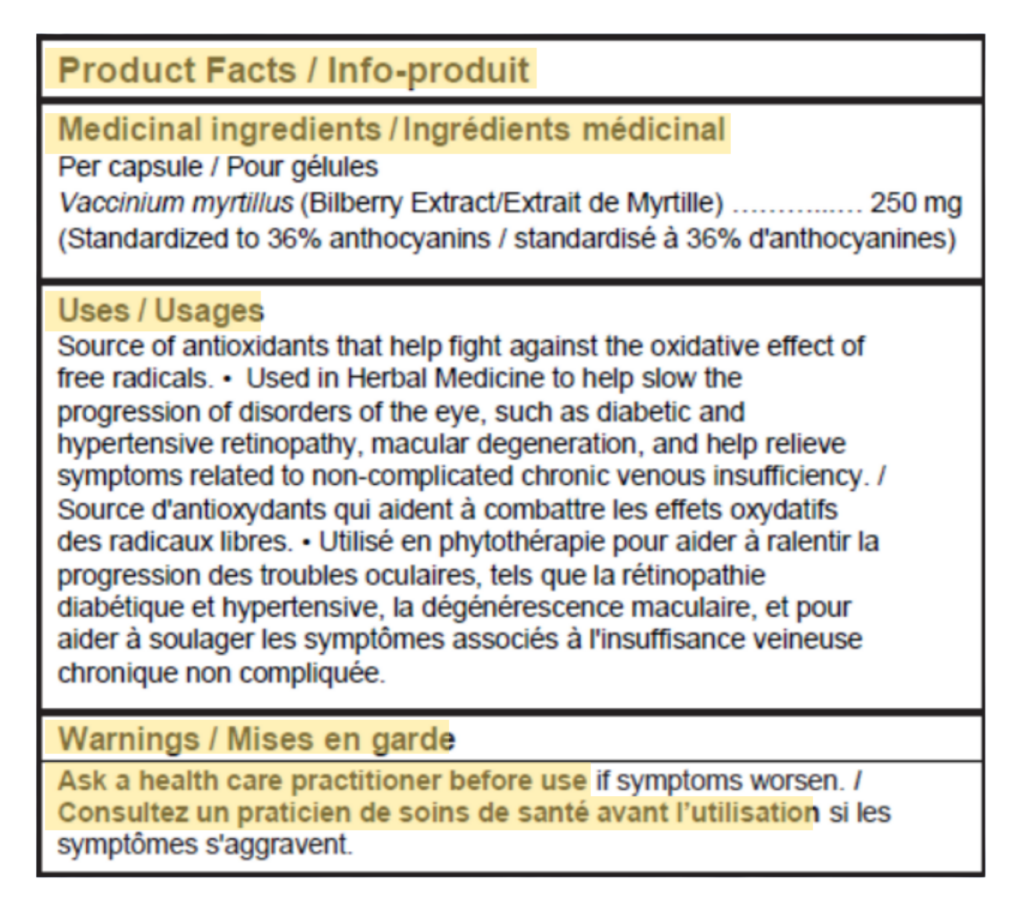
Bilingual Labelling Requirements
The PFT content must be presented in both English and French to ensure accessibility for all Canadian consumers, in alignment with federal bilingual labelling requirements. To support this, manufacturers may either use a single combined table that integrates both languages or present two separate tables, one for each language. A combined table can reduce space constraints by avoiding duplication, while separate tables offer clearer language separation but require more label space.
In addition to federal requirements, Quebec imposes stricter obligations under the Charter of the French Language. All product-related information must appear in French at least as prominently as any other language, and this extends beyond the NHP-specific requirements to include marketing and promotional materials. Manufacturers must therefore assess both federal and provincial requirements to ensure full compliance across all Canadian markets.
Inner and Outer NHP Labelling Requirements
Inner and outer labels may contain different information depending on the packaging configuration, and each has distinct regulatory roles under the Natural Health Products Regulations.
Core Information For Inner and Outer Labels
These elements must appear on the principal display panel of both labels:
- Brand name
- NPN or DIN-HM
- Dosage form (e.g., tablet, capsule, liquid)
- Net quantity (weight, volume, or number of units)
- “Sterile” designation, if applicable
Outer Label (or Inner Label When No Outer Label Is Present)
This is where the full regulatory content is primarily housed, including the Product Facts Table.
Must include:
- PFT, bilingual or separate English/French tables
- Name of product licence holder (or importer, if applicable)
- Recommended route of administration
- Lot number
- Expiry date
Inner Label (When Both Inner and Outer Labels Are Present)
The inner label does not require the full PFT, but must retain key identifying information. Must include (at minimum):
- Brand name
- NPN or DIN-HM
- Dosage form
- Net quantity
Comparison of Previous and Updated Labelling Requirements
| Feature | Previous Regulations | Updated Regulations |
| Format | Varied among brands | Standardized Product Facts Table |
| Clarity | Often technical and ambiguous | Clear and concise language |
| Allergen Disclosure | Not always provided | Mandatory allergen labelling |
| Readability | No specific font-size requirements, not a structured layout | Requirements for font-size and spacing, structured layout |
| Bilingual Requirements | Mandatory English & French labelling | Mandatory English & French labelling |
Historically, product labels varied significantly in format, structure, and presentation, making it difficult for consumers to quickly locate critical product information. Differences in font sizes, layouts, and terminology between brands often resulted in confusion, requiring consumers to spend additional time searching for essential details such as dosage instructions, warnings, and ingredient lists. This lack of consistency not only complicated decision-making but also increased the risk of misuse, particularly for individuals with limited health literacy or visual impairment.
Additionally, the revised regulations introduce a standardized and user-friendly format that ensures all NHP labels present information in a clear and consistent manner. By mandating uniform formatting across all brands, the updated requirements help consumers quickly and easily identify key details, reducing the time required to find essential product information. The introduction of the Product Facts Table plays a significant role in this improvement by organizing information in a structured and predictable way, mirroring the labelling approach used for non-prescription drugs.
Significance of These Changes for Consumers and Industry Stakeholders
The updated labelling requirements for NHPs mark a significant step in enhancing consumer protection. Many consumers struggle to interpret complex labels, leading to misuse, accidental overdoses, or allergic reactions. By introducing standardized, PLL, Health Canada ensures that essential product information is clear, reducing health risks and improving public safety.

For industry stakeholders, these changes enhance consumer trust, reinforce brand credibility, and help businesses avoid penalties, product recalls, and enforcement actions. Aligning with global labelling standards also strengthens the marketability of Canadian products. Additionally, clearer labels reduce consumer confusion, product misuse, and complaints. While adapting to these updates requires investment in redesign and training, businesses that comply will improve customer satisfaction, strengthen their reputation, and ensure long-term success in a competitive market.
Compliance Deadlines for Industry Participants
Health Canada has revised the compliance deadlines for the new labelling regulations for NHPs. Under the latest update, all NHPs must fully comply with the PLL guidelines by June 22, 2028.
Guidelines for Achieving Compliance with the Updated Regulations
To ensure adherence to Health Canada’s revised labelling standards, businesses are advised to undertake the following measures:
Review Health Canada’s Updated Guidelines
Familiarize yourself with the latest regulatory requirements, including the mandatory Product Facts Table (PFT), font specifications, bilingual labelling rules, and allergen disclosure requirements. Ensure your team understands the scope of these changes and their impact on product labels.
Revise Product Packaging and Labels
Update all labels and packaging to meet the new readability, formatting, and allergen disclosure standards. Ensure that essential information, such as active ingredients, dosage, warnings, and expiry dates, follows the standardized format. Labels must use a sans-serif font in a specific size and be printed in a single color equivalent to 100% solid black.
Implement Bilingual Labelling Requirements
Ensure that all required product information is available in both English and French. Manufacturers can choose between a hybrid table that integrates both languages or two separate tables for each language. Businesses operating in Quebec must also comply with provincial regulations, which may require additional translations beyond federal requirements.
Conduct Internal Compliance Audits
Regularly review and assess product labels to verify compliance with the updated Health Canada regulations. Pay close attention to allergen disclosure rules, as labels must clearly indicate common food allergens, gluten sources, and added sulphites in the Warnings section of the PFT.
Train Employees on Compliance Requirements
Provide ongoing training to staff involved in labelling, packaging, and regulatory compliance. Employees should understand the importance of standardized labelling, how to apply the new rules, and how to minimize errors that could lead to enforcement actions.
Consult Regulatory Experts and Industry Specialists
Engage regulatory specialists to ensure a smooth transition and avoid costly penalties, product recalls, or restrictions on sales.
While small and medium-sized enterprises may encounter challenges in adapting to these changes due to redesign costs, training expenditures, and production updates, proactive investment in compliance will prevent costly legal and regulatory complications in the long term.
Learn more about NHP regulations in the Natural Health Product Regulations and NPN Licensing in Canada blog post by SNI.
Regulatory Oversight and Enforcement by Health Canada

Health Canada ensures compliance with the Natural Health Product Regulations through a robust and multifaceted enforcement strategy. This approach integrates various key measures to safeguard public health and maintain regulatory integrity. When formulating a market entry strategy and establishing control checkpoints, it is essential to consider the following critical factors:
1. Routine Audits and Inspections:
Health Canada conducts regular assessments of businesses involved in the manufacturing, packaging, labelling, importing, storing, distributing, selling, and advertising of NHPs. These inspections ensure adherence to the Natural Health Products Regulations and the Food and Drugs Act (FDA).
2. Product Evaluation:
Systematic reviews of NHP packaging and labelling are performed to verify accuracy and compliance with regulatory standards. This process ensures that labels provide clear, consistent, and legible information to consumers, aligning with established guidelines.
3. Enforcement Actions:
In cases of non-compliance, Health Canada has the authority to implement various enforcement actions, including:
- Licence Suspension or Cancellation: Health Canada can suspend or cancel product and site licences for NHPs that do not comply with regulatory requirements.
- Stop-Sale Directives: Issuing orders to halt the sale of non-compliant products.
- Product Seizures: Confiscating products that pose health risks or violate regulations.
- Voluntary Recalls: Requesting companies to recall products that are found to be non-compliant or hazardous.
- Public Advisories: Issuing alerts and advisories to inform the public about safety concerns related to specific NHPs.
4. Educational Support and Resources:
Health Canada provides industry stakeholders with guidance documents, workshops, and other resources to facilitate compliance with regulations. These initiatives aim to promote a clear understanding of regulatory requirements and support businesses in achieving compliance.
Final Remarks
The introduction of the PLL regulations represents a significant step toward improving consumer safety and ensuring clarity in NHP labelling across Canada. By mandating a standardized Product Facts Table, requiring clear allergen disclosures, and enforcing strict readability standards, Health Canada has taken proactive measures to reduce confusion and enhance transparency in the NHP sector.
For consumers, these changes mean greater confidence in understanding product details, reducing the risk of misuse or adverse reactions. For industry stakeholders, compliance with these regulations not only ensures adherence to legal requirements but also fosters consumer trust, enhances brand credibility, and aligns Canadian NHP labelling standards with global best practices.
While adapting to these new requirements may pose challenges for some businesses, the long-term benefits of improved safety, regulatory clarity, and consumer satisfaction outweigh the initial investment in compliance. As the June 22, 2028, deadline approaches, industry participants must take proactive steps to update their labels, train their teams, and implement best practices to meet the new standards.

SNI’s Consulting Services for NHP Compliance and New Labelling Requirements Alignment
Looking to bring a NHP to market in Canada? SNI’s Regulatory Affairs team supports end to end NHP licensing and compliance, with a strong focus on meeting Health Canada’s PLL requirements. From product classification and licence application strategy to PFT development and bilingual label compliance, we ensure your product is aligned with current regulatory expectations from the outset.
Whether you are pursuing a streamlined monograph pathway or a more complex submission, we help structure your application to reduce review delays and support efficient approval. Our team integrates regulatory, labelling, and compliance considerations early in development, ensuring your product is not only licensed, but market ready.
⬇️ Send us a request for support or an introductory call
FAQ
What is PLL for NHPs?
Plain Language Labelling, or PLL, refers to Health Canada’s updated requirements for how information appears on NHP labels. The goal is to ensure that consumers can easily understand key details like dosage, ingredients, warnings, and contact information by presenting it in a standardized format across all NHPs sold in Canada. All product information must now appear in a standardized Product Facts Table using clear language, readable fonts, strong contrast, and bilingual formatting.
Who needs to comply with the new labelling requirements and by when?
All companies that sell NHPs in Canada must comply with the new regulations. Products licensed before June 21, 2025, must transition to the updated labelling standards by June 22, 2028. Products licensed after June 21, 2025, may temporarily use the older labelling format but will also need to update by the 2028 deadline.
What changes are required for label formatting and design?
Labels must include a bilingual Product Facts Table that clearly outlines the product’s medicinal and non-medicinal ingredients, recommended use, dosage instructions, warnings, storage conditions, and contact details. The label must use a sans-serif font in a minimum size, with high-contrast black text on a white or nearly white background. Headings must be bolded, and sections must be clearly separated to help consumers find information quickly. For more information on labelling specifications and formatting requirements, please visit Health Canada’s NHP labelling regulations.
How must allergens be declared under the new regulations?
Labels must clearly disclose the presence of common allergens such as milk, nuts, wheat, soy, and shellfish, along with gluten sources and added sulphites. All priority food allergen information must be listed in the Warnings section of the Product Facts Table in a way that is easy to find. If an ingredient is derived from an allergenic source, it must be identified accordingly to help consumers avoid potential reactions.
Are there any exceptions to the full labelling requirements?
Yes, certain products may qualify for labelling flexibilities. These include small packages, short-term use items, and products with localized effects, such as topical creams. In these cases, manufacturers may use fold-out labels, inserts, or provide digital access to the full Product Facts Table, as long as the information is available and easy for the consumer to access at the time of use or purchase. For more information on flexibilities, please visit Health Canada’s NHP labelling regulations.
✷ The content on this website, including information presented in this post, is provided for general informational purposes only and does not constitute legal, regulatory, or professional advice. While efforts are made to ensure accuracy, laws and regulations vary by jurisdiction and may change over time. Readers should not rely on this information as a substitute for advice from qualified legal or regulatory professionals. We disclaim any liability for actions taken based on this content, and users are encouraged to seek guidance specific to their circumstances.




