On April 23, 2026, Health Canada has formally confirmed the transition of sports electrolyte products from the Natural Health Product (NHP) framework to the food regulatory framework. This update, issued by the Health Canada Food and Nutrition Directorate in collaboration with the Natural and Non-prescription Health Products Directorate (NNHPD), reflects a broader regulatory shift toward aligning oversight with product format, representation, and consumer perception.
For industry stakeholders, this change alters the regulatory pathway for a category that has historically operated at the interface between foods and NHPs. While the transition reduces reliance on pre-market licensing, it introduces new compliance obligations under the Food and Drugs Act and the Food and Drug Regulations, including requirements applicable to supplemented foods where relevant.
- What Are Sports Electrolyte Products?
- Distinction Between Sports Electrolyte Products and Oral Rehydration Solutions
- Regulatory Rationale for the Transition of Sports Electrolyte Products from NHP to Food Regulations
- Sport Electrolyte Product Transition Timeline and Compliance Expectations
- Regulatory Requirements Under the Food Framework
- Labelling and Claim Considerations for Sports Electrolyte Products
- Strategic Considerations for Industry Introducing Electrolyte Products
- Final Remarks
What Are Sports Electrolyte Products?
Sports electrolyte products are defined by both their format and their intended use. Health Canada has emphasized that these products are typically presented in conventional food formats and are consumed in a manner consistent with other beverages and drink mixes available on the market.
These products are typically sold as ready-to-drink beverages, powders or concentrates to be mixed with water, and effervescent tablets that dissolve before consumption. They are marketed for hydration during and after exercise and for electrolyte replacement, a positioning that aligns closer with conventional food use rather than therapeutic intent.
Distinction Between Sports Electrolyte Products and Oral Rehydration Solutions

A critical element of this transition is the explicit distinction between sports electrolyte products and Oral Rehydration Solutions. Oral Rehydration Solutions are formulated for the treatment of dehydration associated with clinical conditions such as diarrhea or vomiting and will remain regulated under the NHP framework.
Products that currently carry both sports electrolyte positioning and Oral Rehydration Solution claims will be required to make a clear regulatory determination. Manufacturers and responsible parties of such products must either remove sports-related claims in order to remain within the NHP framework as an Oral Rehydration Solution or remove therapeutic claims and transition fully into the food regulatory framework. This distinction is rooted in intended product use, risk profile, and formulation.
Regulatory Rationale for the Transition of Sports Electrolyte Products from NHP to Food Regulations
The transition of sports electrolyte products into the food framework reflects principles outlined in Health Canada’s guidance on classification at the food and NHP interface. The central premise is that products in food formats, marketed and consumed as foods, should be regulated as such.
This reclassification is intended to improve consistency across similar product categories and reduce discrepancies in labelling requirements that may otherwise create confusion for consumers. It also supports a more streamlined regulatory environment by aligning oversight with established food regulations, thereby reducing administrative complexity for both industry and regulators. At a broader level, the shift provides clearer and more predictable information to consumers, enabling more informed purchasing decisions.
Sport Electrolyte Product Transition Timeline and Compliance Expectations
Health Canada has established a transition period that allows existing licence holders to adapt to the new regulatory framework. Companies holding Natural Product Numbers (NPN) for sports electrolyte products are encouraged to transition by December 31, 2027, in order to be considered compliant under the food regulatory framework.

During this period, products may continue to be sold under their existing authorizations. However, this should not be interpreted as a passive allowance. Rather, it represents a defined window for companies to undertake the necessary regulatory, formulation, and labelling changes required for compliance under the Food and Drug Regulations.
Where additional time is required, licence holders have the option to submit a transition plan to the NNHPD for consideration. Health Canada has also indicated that it will engage directly with affected stakeholders to support the transition process.
Regulatory Requirements Under the Food Framework
Once transitioned, sports electrolyte products will be regulated as foods and must comply with the Food and Drugs Act and the Food and Drug Regulations. Unlike NHPs, these products will not require pre-market licensing. Instead, the responsible party must ensure compliance before sale, with oversight occurring primarily through post-market compliance and enforcement.
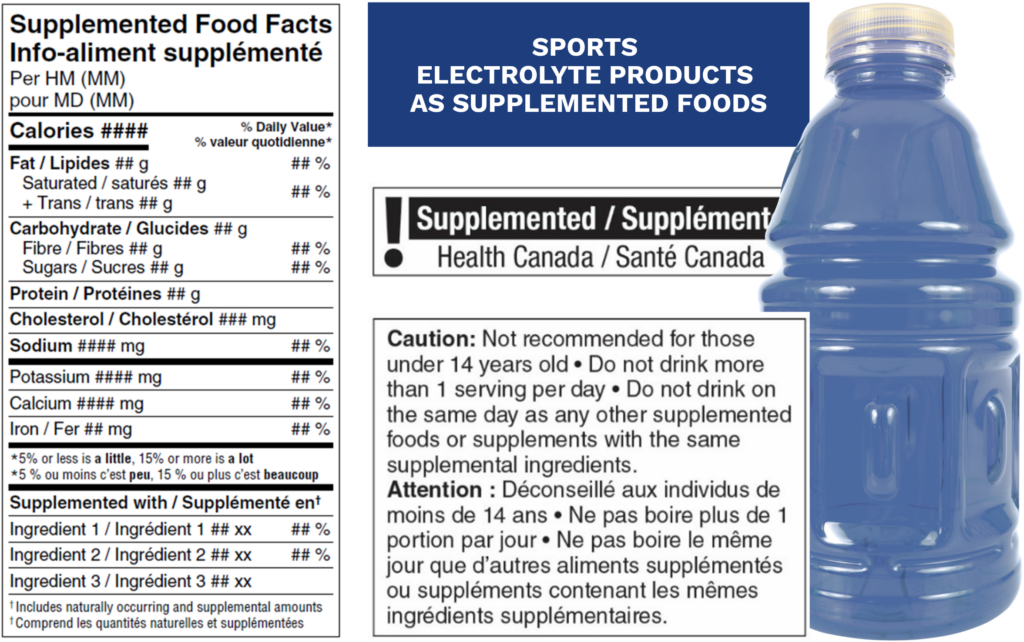
In many cases, sports electrolyte products will be classified as “supplemented foods” due to the addition of ingredients such as vitamins, minerals, amino acids, or other supplemental ingredients. Food products containing supplemental ingredients and appearing on the list of permitted supplemented food categories must comply with category-specific regulatory requirements, including adherence to permitted ingredient lists, maximum levels of use, and prescribed labelling elements. This includes the use of the Supplemented Food Facts table (SFFt) and any required cautionary statements or identifiers. For more information on supplemented foods and their regulations, please read: Canada’s Supplemented Food Labelling and Regulations
Labelling and Claim Considerations for Sports Electrolyte Products
Moving to the food framework changes how labelling and claims are regulated. Products must meet food labelling requirements, including the appropriate Nutrition Facts table (NFt) or SFFt, a complete ingredient list, allergen declarations, and bilingual labelling. Where required, front-of-package nutrition symbols must also appear.
Claims also need reassessment. All express and implied claims must comply with the Food and Drugs Act and the Food and Drug Regulations, and there is no monograph-based pathway as under the NHP framework. As a result, claims must be substantiated, accurate, and not misleading, often requiring more conservative, evidence-driven marketing language.
Learn more about food regulations in in our articles:
- How to Launch a Compliant Food Product in Canada: Understanding Canadian Food Labelling Regulations in 2025
- Canadian Bilingual Food Labelling Requirements: What You Need to Know
- Nutrition Facts Table Regulations in Canada: How to Meet the Requirements
- How to Comply with New Front-of-Package (FOP) Nutrition Label Requirements for Foods in Canada
Strategic Considerations for Industry Introducing Electrolyte Products
For existing NPN holders, the transition is a substantive regulatory shift. Companies should confirm whether current formulations fit within conventional or supplemented food permissions and identify any needed reformulation. Labelling must be updated to meet food standards, and claims must be reassessed for compliance under the new framework.
The transition can also streamline market access. Removing pre-market licensing may accelerate launches, provided compliance is in place before sale. At the same time, it shifts more responsibility to companies to confirm all requirements are met prior to commercialization.

Need help navigating the transition? SNI supports reformulation, label updates, and regulatory strategy so you can move to the food framework with confidence. With deep expertise in food and NHP regulations, we deliver practical, tailored guidance to keep your product compliant and launch-ready.
Final Remarks
The reclassification of sports electrolyte products as foods reflects a deliberate policy direction by Health Canada toward greater regulatory clarity and consistency. By aligning classification with product format and consumer use, the framework becomes more predictable and coherent.
For industry, the implications are twofold. While the transition removes the pre-market licensing burden associated with the NHP framework, it introduces a distinct set of compliance obligations under the Food and Drug Regulations that require deliberate planning and execution. Organizations that take a proactive, structured approach to this transition will be better positioned to maintain compliance while capitalizing on the operational efficiencies of the food framework.

SNI Services for Regulated Products
Planning to transition your sports electrolyte product into the Canadian food framework requires a structured and well-informed approach. SNI’s Regulatory Affairs team supports companies across consumer-packaged goods and adjacent regulated product categories with classification assessments, supplemented food compliance, labelling strategy, and claims substantiation. From initial gap analysis through to commercialization, our team ensures that regulatory transitions are managed efficiently and in full alignment with current Health Canada expectations.
⬇️ Send us a request to learn more about our services
The content on this website, including information presented in this post, is provided for general informational purposes only and does not constitute legal, regulatory, or professional advice. While efforts are made to ensure accuracy, laws and regulations vary by jurisdiction and may change over time. Readers should not rely on this information as a substitute for advice from qualified legal or regulatory professionals. We disclaim any liability for actions taken based on this content, and users are encouraged to seek guidance specific to their circumstances.




