On March 27, SNI attended an invited session on NNHPD’s Updated Proposal for NHP Cost Recovery
We want to share some updates regarding the ongoing cost recovery initiative that has been imposed on the NHP sector.
Last year, Health Canada proposed a new fee structure for Natural Health Products (NHPs) which left industry with many questions and concerns (see our previous blogs here and here).
Health Canada has now released a revised fee proposal for NHPs, offering lower fees and eliminating the Class III Novel fee category.
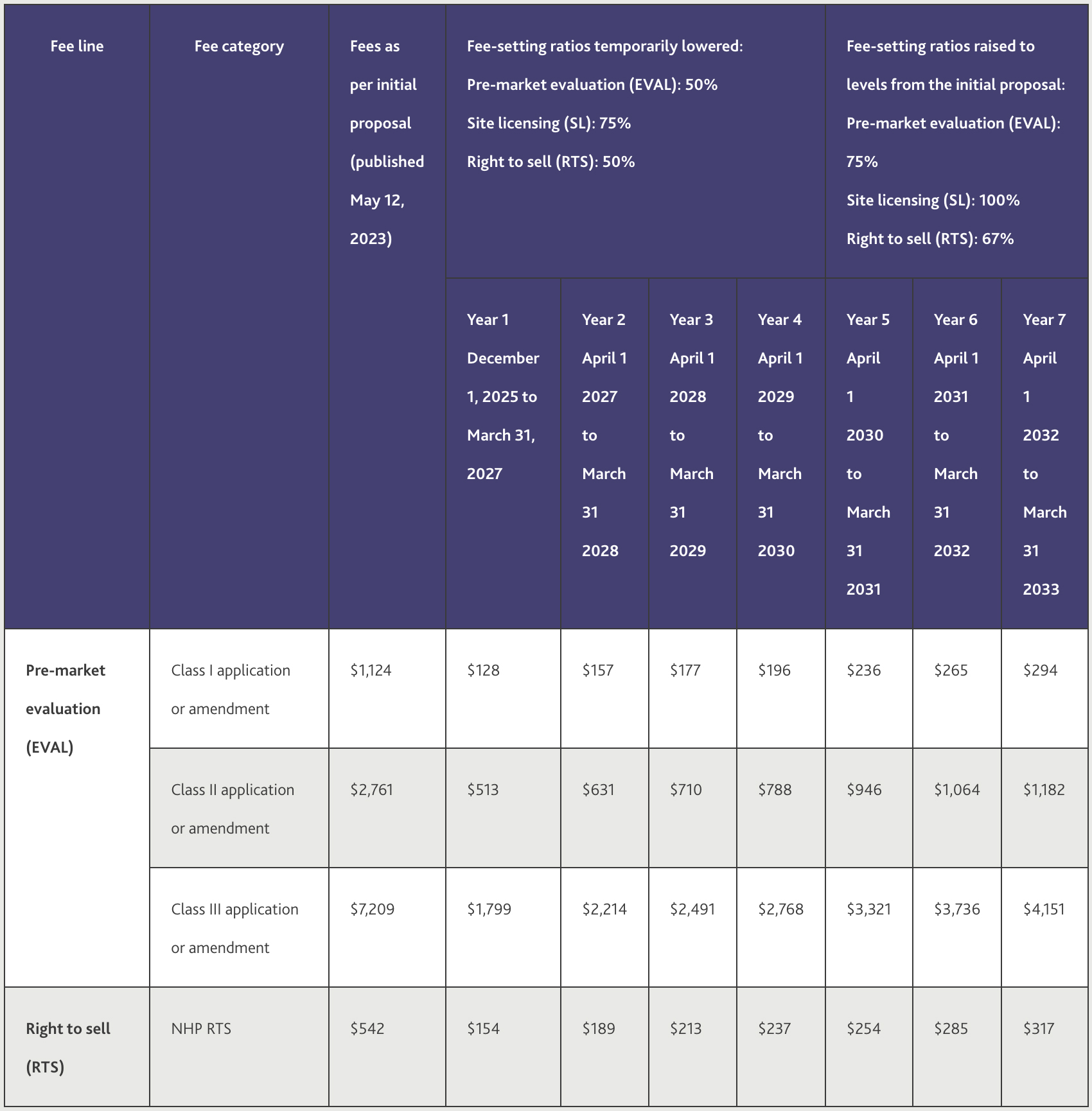
THESE FEES WOULD BE PHASED-IN OVER SEVEN YEARS STARTING FROM DECEMBER 1, 2025
Concerns continue regarding the lack of revisions to small business mitigation measures. While businesses meeting specific criteria may qualify for fee reductions, some stakeholders believe that the overall fee structure still presents challenges, especially for small companies.
After this revised proposal, questions remain about how Health Canada calculated the costs, their alignment with other initiatives, and the potential impact on the NHP industry within Canada.

PROPOSAL OVERVIEW
Health Canada's proposal encompasses fees for all NHP applications and amendments, categorized by the class of Product Licence Application (PLA), as outlined in Table 1 below. Notably, Health Canada has addressed industry concerns regarding the proposed Novel Class III category, which was initially set at $58,332.
The proposed fees are lower overall and phased in over a 7-year approach.
NHP SITE LICENSES
Site license fees will be imposed for the review of applications and amendments, as detailed in Table 2 below.
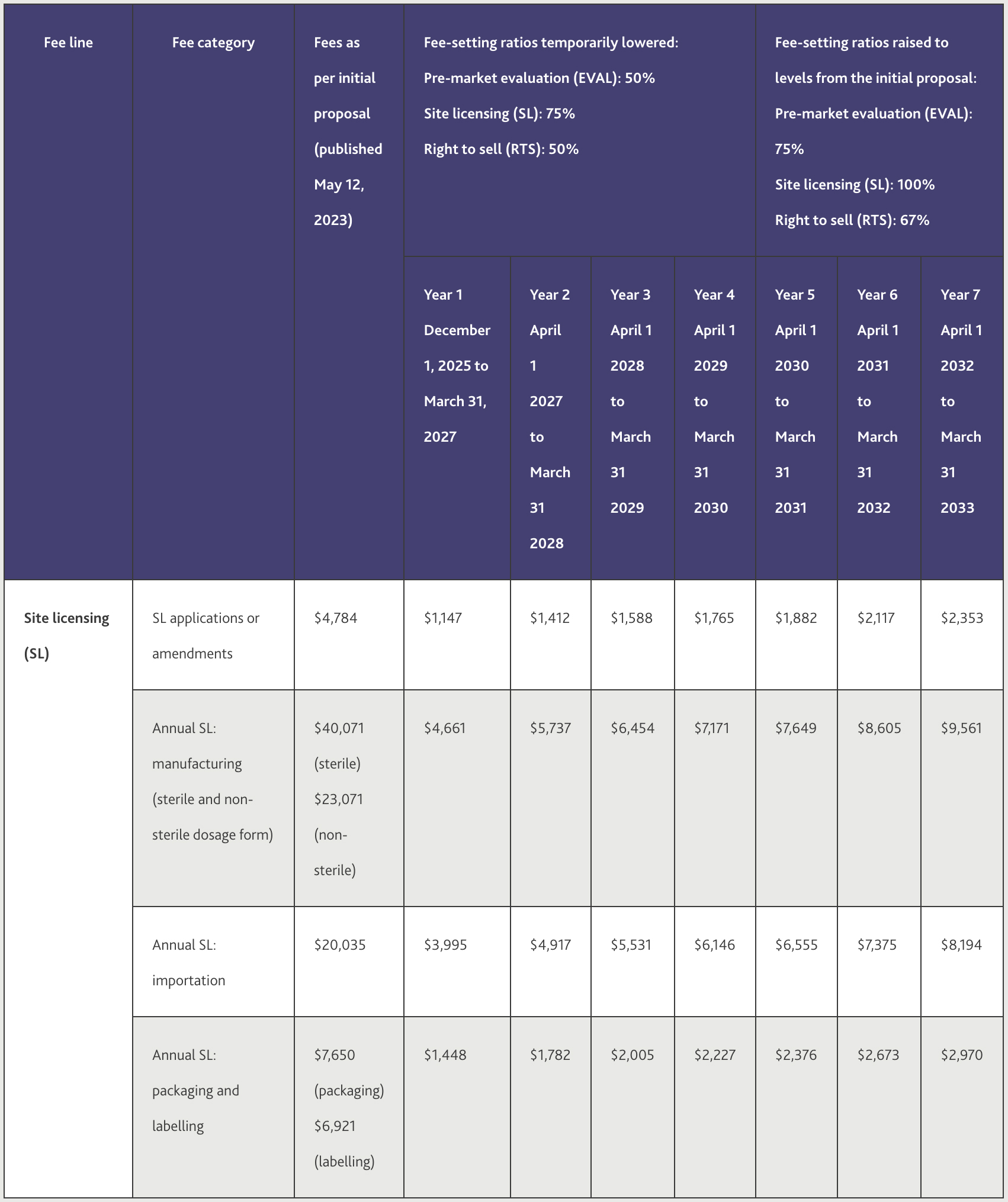
Health Canada notes that these cost reductions result in fees lowered by 38% to 72% (depending on the fee) once fully implemented.
There are discounts for companies qualifying as a small business, in an attempt to reduce undue burden. Health Canada has opened a 60-day consultation, you can have your say!
The online consultation ran from March 27 to May 25, 2024 and is now closed.
Team SNI will continue to actively monitor the situation and provide updates as they arise. If you have any questions about these regulatory changes, please reach out today.
Contact us at info@sourcenutra.com for all your industry inquiries.




