Creatine has become a popular household staple around the world, widely recognized for its role in supporting athletic performance and lean muscle development. Increased consumer demand for creatine has driven significant innovation, leading brands to incorporate creatine into a growing range of product formats, including beverages, gummies, bars, and even dairy alternatives. Wherever you look, creatine is there.
Although creatine is a well-established ingredient, manufacturers must still ensure proper regulatory planning and confirm whether its addition to a target product is permissible in the markets they intend to enter. In some jurisdictions, creatine may be used in certain food formats, while in others its use may be limited to supplement-type products or a narrower range of applications. As a result, early classification and pathway planning remain critical to avoiding unnecessary reformulation, relabelling, or launch delays.
In this article, we explore how creatine and creatine-based products are regulated in Canada, providing an overview of the available pathways, key considerations, licensing requirements, and the most common causes of market-entry delays.
- How is Creatine Regulated in Canada
- Confirming Creatine Finished Product Fit Within the Canadian NHP Pathway
- Selecting the Fastest NHP Licensing Pathway for Creatine in Canada
- Planning for the Most Common Causes of Licensing Delays for Creatine NHPs
- Final Remarks: Building the Fastest Creatine Market Entry Pathway for Canada
- FAQ
How is Creatine Regulated in Canada
The first step in bringing a creatine product into Canada is determining which regulatory framework applies. Even where the base formula remains unchanged, the available market-entry pathway may differ depending on ingredient permissibility, proposed claims, and the finished product’s commercial presentation. This assessment typically begins by determining whether the product can proceed through an established Natural Health Product (NHP) pathway or whether a more complex food-based strategy will need to be explored.
Creatine as a Natural Health Product (NHP)
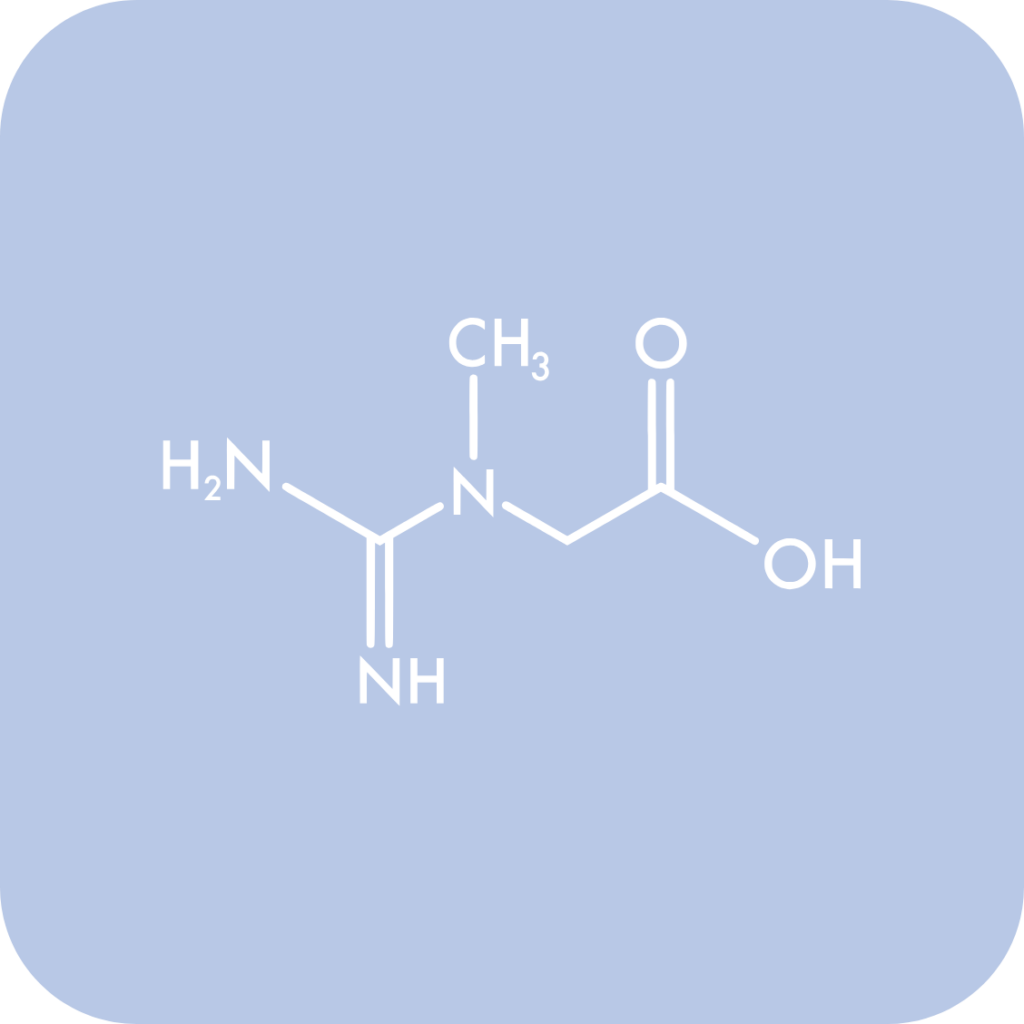
In Canada, creatine is regulated as an NHP ingredient under the Natural Health Products Regulations. This is currently the most established market-entry pathway, with the permitted use in NHP dosage formats such as powders, gummies, and tablets, while liquids and solutions remain excluded due to insufficient finished product stability evidence.
Creatine is currently supported under both the single-ingredient Creatine Monohydrate monograph and, where formulation context permits, the Workout Supplements monograph, with permitted daily doses generally ranging from 3 g to 20 g, depending on the directions of use.
Creatine as a Food
Although the NHP route is currently the most established pathway for creatine products entering Canada, a food positioning may still be explored where commercially justified. However, because creatine is not currently permitted for use in conventional or supplemented foods, there is no standard food route available at the outset.
A food-based strategy is therefore typically limited to exceptional pre-market assessment pathways, where creatine must be reviewed in the context of its proposed use. Only where sufficient safety and use-specific evidence is available may the ingredient be deemed non-novel or considered for addition to the List of Permitted Supplemental Ingredients. Because this route introduces additional regulatory uncertainty and longer timelines, early feasibility assessment is critical to determine whether the existing product can proceed as designed or whether reformulation under the NHP pathway would be the more efficient route to market.

Confirming Creatine Finished Product Fit Within the Canadian NHP Pathway
Once the perceived product category has been established, the next step is confirming whether the finished imported product fully fits within the selected Canadian regulatory pathway. Because the food route is more complex and less predictable, the preferred route to market is often through the NHP pathway. As such, the focus typically shifts to evaluating whether the product is appropriately aligned with the NHP category. At this stage, the assessment moves from high‑level categorization into a detailed gap review of the product’s format, representation, recommended use, claims, and overall commercial context. This gap review is conducted to confirm whether the product can proceed through the intended pathway without triggering additional regulatory considerations.
Assessing Product Format Against the NHP Pathway
For products containing creatine, product format is often the first point of gap assessment against Canadian regulations. Dosage‑style presentations typically align more readily with the NHP pathway, while food‑like formats often prompt reassessment of whether the product can enter Canada as‑is or whether an alternative regulatory pathway, pre‑market review, or reformulation will be required.
In some cases, products that initially appear food‑like may, following a thorough assessment, ultimately be confirmed as acceptable under the NHP framework. Given this nuance, a detailed regulatory gap assessment is often required before advancing the selected pathway
Assessing Claims and Product Context for NHP Fit
Once product format is confirmed to be acceptable within the NHP category, product claims and overall commercial context must also be assessed for NHP compliance. Even where the format itself meets the NHP definition, the licensing pathway may become more complex if the product’s presentation, marketing context, or claims fall outside the scope supported by applicable Health Canada monographs. That said, where product claims and overall representations remain permissible under the Natural Health Products Regulations, the product may still proceed through an established NHP route to market, albeit via a different licensing pathway than a monograph‑only submission.

Selecting the Fastest NHP Licensing Pathway for Creatine in Canada
Once the finished product has been confirmed to fit within the Canadian NHP pathway, the next step is selecting the most efficient licensing route. At this stage, the objective is to determine how the Product Licence Application (PLA) can be structured to support the fastest possible review while remaining fully aligned with the product’s ingredient form, dose, claims, and supporting monograph strategy.
Confirming the Natural Product Number (NPN) Requirement
In all cases where the product is classified as an NHP, a Natural Product Number (NPN) is required before the product can be legally sold in Canada. The NPN confirms that Health Canada has reviewed and authorized the product for sale, including its medicinal ingredients, recommended use or purpose, dose, and supporting claims. Obtaining the NPN is one of the first major regulatory milestones in the Canadian market-entry pathway, as the product cannot be commercially distributed until the licence is issued.
Proceeding Through a Class I or Class II Monograph-Based PLA

Where the finished product fully aligns with the applicable Creatine Monohydrate or Workout Supplements monograph, the most efficient route to market is typically a Class I PLA, as the submission can rely on a single monograph attestation to support the ingredient, dose, claims, and quality parameters.
However, where creatine is combined with medicinal ingredients supported under a separate NHP monograph, the application generally shifts to a Class II PLA, since Health Canada must assess the combined monograph attestations together. While still considered an efficient route, this additional review step typically results in longer timelines than a single-monograph Class I submission.
Proceeding Through a Class III Evidence-Based PLA
Where the finished product cannot fully align with the applicable creatine monograph, the next step is determining whether the product can proceed through a Class III PLA. This typically applies when the ingredient specifications, recommended dose, claims, medicinal ingredient combinations, or creatine form fall outside the published monograph scope.
In these situations, the applicant must submit a product-specific evidence package to justify each non-monographed element. This evidence must demonstrate that the proposed ingredient use, dose, and claims remain safe, efficacious, and suitable for licensing despite falling outside the standard monograph pathway. Because this route introduces the highest evidence burden, it also tends to carry the longest review timelines.

Planning for the Most Common Causes of Licensing Delays for Creatine NHPs
Once the licensing pathway has been selected, the next priority is identifying the issues most likely to delay Health Canada review. Addressing these risks early can reduce deficiency cycles and help keep Canadian launch timelines on track.
Preventing Delays Caused by Unsupported Creatine Sources
A common creatine-specific delay risk arises where the finished product uses a creatine source that falls outside the supported monograph scope. While Health Canada’s most established pathways are built around Creatine Monohydrate, alternate sources such as creatine hydrochloride, buffered creatine, creatine nitrate, or proprietary salts may not fit within the same streamlined route.
This commonly occurs where international products are filed under the assumption that all creatine sources are treated equivalently in Canada. In practice, unsupported sources often shift the submission into a Class III PLA, which extends licensing timelines.
Preventing Delays Caused by Weak Supporting Evidence

Where the submission proceeds through a Class III PLA, delays commonly arise where the evidence package does not adequately support the proposed non-monographed claims, dose range, or ingredient combinations.
For example, if a creatine product is positioned with a cognitive performance or healthy aging claim, but the evidence relies only on sports performance studies in young athletic populations, Health Canada may determine that the proposed use is not adequately supported. Multiple weak evidence points can quickly lead to deficiency notices, extended review cycles, or the need to refile.
Preventing Delays Caused by Importation and Release Readiness
Even after licensing is complete, delays can still arise where the Canadian importation and commercial release strategy has not been established. Under the standard NHP route, finished products must be imported by a site-licensed importer responsible for release, recordkeeping, and Good Manufacturing Practices (GMP) compliance prior to sale.
For foreign brands, this commonly occurs where the NPN is secured before confirming the Canadian importer of record, site licence scope, or release procedures. In these cases, products may be licensed but still unable to enter the market on the planned launch timeline.
Final Remarks: Building the Fastest Creatine Market Entry Pathway for Canada
For most international creatine products entering Canada, the most efficient route to market begins with early confirmation that the finished product fits within the NHP framework and aligns with the applicable Creatine Monohydrate or Workout Supplements monograph pathway. From there, the speed of licensing is largely determined by how well the product’s ingredient form, recommended dose, claims, and supporting evidence align with Health Canada’s established expectations.
Where these considerations are assessed early, brands can often avoid unnecessary reformulation, repeated deficiency cycles, and launch delays. By contrast, pathway assumptions made too late, particularly around unsupported creatine forms, non-monographed claims, or post-licensing importation readiness, can materially slow Canadian commercialization timelines.

Bringing Your Creatine Products to Canada with SNI
At SNI, we support international brands with the full Canadian market-entry pathway for creatine and sports nutrition products, from initial product classification and pathway fit-gap assessments through to PLA preparation, evidence strategy, NPN licensing, importer readiness, and post-licensing GMP support.
For brands entering Canada with existing monohydrate powders, pre-workouts, gummies, or proprietary performance formulations, our team helps determine the most efficient licensing route while proactively identifying the regulatory and operational issues most likely to delay launch. Where the existing international product does not fully align with Health Canada’s monograph pathways, we also support evidence-driven Class III strategies and reformulation planning to protect launch timelines.
If your team is preparing to bring a creatine product into Canada, early pathway assessment is often the difference between a streamlined NPN review and months of avoidable delay.
⬇️ Send us a request to learn more about our services
FAQ
The licensing timeline depends on the selected PLA class. A Class I PLA aligned with the Creatine Monohydrate or Workout Supplements monograph is typically the fastest route (60 days), while Class II (90 days) and especially Class III (310+ days) applications carry progressively longer review timelines due to increased assessment complexity and evidence requirements
In some cases, yes, but this depends on whether the existing ingredient form, recommended dose, claims, and dosage format align with Canadian NHP expectations. U.S. products using alternate creatine salts, liquid presentations, or broader performance and lifestyle claims often require pathway reassessment, relabelling, or reformulation before Canadian sale.
Yes. Even after the product receives an NPN, creatine products must be brought into Canada by a site-licensed importer that is responsible for release, recordkeeping, and GMP compliance prior to sale.
No, even where the finished formula is acceptable, labels often require Canadian-specific updates to align with approved claims, recommended use, dose instructions, cautionary statements, and bilingual requirements where applicable.
Post-licensing changes may require notification, amendment, or full re-licensing, depending on the nature of the update. Changes to the creatine ingredient form, recommended dose, claims, or medicinal ingredient combinations should always be assessed before implementation to avoid post-market non-compliance.
✷ The content on this website, including information presented in this post, is provided for general informational purposes only and does not constitute legal, regulatory, or professional advice. While efforts are made to ensure accuracy, laws and regulations vary by jurisdiction and may change over time. Readers should not rely on this information as a substitute for advice from qualified legal or regulatory professionals. We disclaim any liability for actions taken based on this content, and users are encouraged to seek guidance specific to their circumstances.




