We previously mentioned in a recent blog post that new labelling requirements were coming soon for Natural Heath Products (NHPs).
Well that day has arrived!
On July 6, 2022, Health Canada brought in the requirement to have a Product Facts Table (PFT) on all Natural Health Products, as well as some other adjustments to the current regulations. Products licenced after July 6th, 2022 will have 3 years to comply with the new labelling requirement and products licenced before July 6th, 2022 will have an additional 3 years for a total of 6 years to comply with the new requirements.
Similar to Food, Drugs and Non-Prescription Drugs, NHPs must now provide an easy-to-read table for consumers with minimum requirements that include:
- Medicinal ingredients
- Uses
- Warnings
- Directions
- Other information (storage requirements)
- Non-medicinal ingredients
- Contact # for Questions
The regulations now require additional labelling for the following:
- food allergens, gluten, added sulphites;
- asthma alert statement; and
- aspartame labelling.
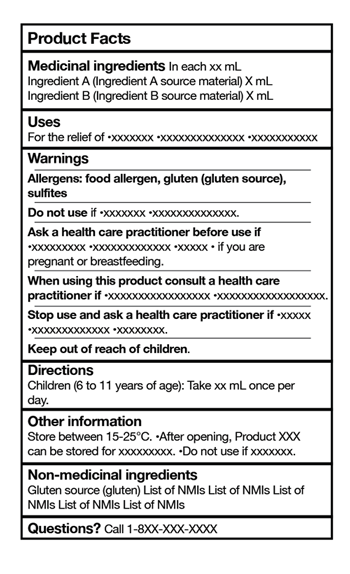
Specific rules on placement of the PFT, the font size, colours that can be used on the label, whether borders within the table can be eliminated to save space on the label for other labelling requirements are also outlined within the updated labelling guidance document.

As with most government regulations and new requirements, the PFT rules can be confusing, with some requiring interpretation by experienced regulatory professionals.
If your business is looking for clarification of the new regulations or any Health Canada regulations relating to foods, NHPs, cosmetics, medical devices, drugs or non-prescription drugs, SNI can help!
Our experienced Creative Services team can also assist with creation of your new PFT compliant label, and any other packaging needs.




